|
ince 1967, Sebia has been a global leader in protein testing using liquid flow separation technology. With a continuous stream of new and innovative products and tests, Sebia now brings this same innovative spirit to the next generation of testing utilizing an advanced separation technique on our capillary systems. We continue to invest in research and development to help automate workflow and increase productivity in the routine clinical laboratory. Whatĺs new at Sebia?
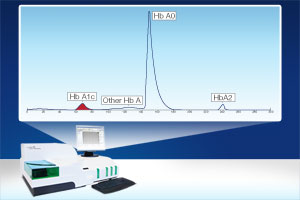
Sebia announces a new generation in HbA1c testing using advanced liquid flow separation technology on the CAPILLARYS 2 Flex Piercing and the MINICAP Flex Piercing platforms. This innovative HbA1c assay provides clear-cut and precise results with no interference from common analytical interfering substances or hemoglobin variants; assuring accurate and clinically significant results. Sebiaĺs assay was developed with the new IFCC and NGSP guidelines and our state-of-the-art instrumentation includes auto flagging of hemoglobin variants and/or beta-thalassemia samples for easy classification and interpretation of HbA1c results.
MINICAPTM Flex Piercing
Sebiaĺs newest instrument, The MINICAP Flex Piercing, offers flexibility, scalability and full automation for low to mid-volume laboratories. The MINICAP Flex Piercing offers whole blood and cap piecing technology for the next generation of HbA1c testing. This small footprint analyzer provides clear-cut and precise results using proven capillary separation technology for both HbA1c and protein testing. Simply place a bar-coded primary tube on the system and walk away; for HbA1c, the whole blood samples are automatically inverted prior to analysis and a continuous access sample wheel allows for the addition of samples at any time. True positive sample ID and full traceability are achieved on this instrument. Menu expansion is being developed to include Hemoglobin, Immunotyping and CDT.
CAPILLARYSTM 2 Flex Piercing
The CAPILLARYSÖ 2 FLEX Piercing instrument is now available for whole blood hemoglobinopathy and thalassemia analysis plus the next generation of HbA1c testing. The system uses automated sample rack inversion, to provide uniform mixing, and cap piercing technology; to allow for no pre-analytical preparation of whole blood primary tube samples. The CAPILLARYS 2 Flex Piercing system provides the fastest hemoglobinopathy and thalassemia assay with up to 38 assays per hour. To provide flexibility and instrument consolidation, the CAPILLARYS 2 Flex Piercing also offers, in addition to hemoglobin and HbA1c testing, a complete test menu to include serum and urine Protein(e)6, Immunotyping and carbohydrate-deficient transferrin (CDT).
| 
